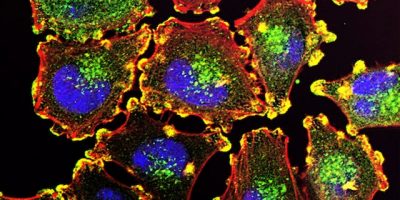
In the quest to unravel the complexities of cancer development, treatment, and prevention, scientists are increasingly exploring cellular metabolism. The realization that metabolic processes are not merely victims of cancer, but active contributors to its development and progression has unveiled an array of potential therapeutic targets. Central to this intricate network of metabolic processes is a coenzyme called nicotinamide adenine dinucleotide, also known as NAD.
The Role of NAD+ in Cellular Metabolism
The NAD molecule is a crucial element in cellular metabolism, as it mediates hundreds of enzymatic reactions. It is critical for processes such as DNA repair, inflammation modulation, circadian rhythm, and the regulation of other metabolic pathways. Importantly, NAD also influences the balance between cell survival and death, a feature that has been heavily implicated in cancer biology.
NAD+ Imbalance and Cancer
Under normal conditions, the concentrations of NAD are tightly controlled to ensure cellular homeostasis. However, research has shown that many types of cancer cells exhibit an abnormal increase in NAD levels. This NAD imbalance provides an environment conducive to cancer progression by favoring the survival of damaged cells, promoting inflammation, and aiding in the evasion of apoptosis, a mechanism that the body uses to eliminate potentially cancerous cells.
This aberrant NAD regulation in cancer cells presents an intriguing metabolic vulnerability that could be exploited for therapeutic benefit. By manipulating the NAD levels, we might be able to affect the survival of cancer cells and possibly halt tumor growth.
Targeting NAD+ Metabolism for Cancer Therapy
The biological importance of NAD has prompted extensive research into the enzymes involved in its metabolism. Two enzymes, in particular, have been the focus of therapeutic targeting: NAMPT and SIRT1.
NAMPT (Nicotinamide phosphoribosyltransferase) is the rate-limiting enzyme in the NAD biosynthesis pathway. In many cancers, NAMPT is overexpressed, leading to increased NAD levels. NAMPT inhibitors have been developed and shown to deplete NAD, leading to the death of cancer cells in preclinical models.
SIRT1 (Sirtuin 1) is a NAD-dependent enzyme that removes acetyl groups from proteins, influencing various cellular processes such as DNA repair and cell survival. Overexpression of SIRT1 has been observed in several types of cancer, where it helps cancer cells evade apoptosis. SIRT1 inhibitors could therefore be a promising avenue to promote cancer cell death and combat tumor growth.
Clinical Trials and Future Directions
Several NAMPT and SIRT1 inhibitors are currently being tested in clinical trials, demonstrating promising initial results. As we delve further into this novel therapy, optimizing efficacy and minimizing side effects will pose a significant challenge. The success of these inhibitors will heavily rely on their ability to specifically target cancer cells, differentiating them from normal cells.
Looking ahead, an in-depth comprehension of NAD metabolism holds the potential to unlock new therapeutic targets. By harnessing an individual’s distinctive metabolic profile, personalized treatments may revolutionize the field of medicine, leading to improved patient outcomes and enhanced quality of life.
NAD+ Provides Hope
The interplay between NAD and cancer provides a fascinating glimpse into the metabolic intricacies of this devastating disease. The abnormalities in NAD levels and the associated enzymes represent a unique metabolic vulnerability in cancer cells that can be potentially exploited for therapeutic gain.
As we delve deeper into understanding these relationships, the hope is that this knowledge will translate into more effective and targeted cancer therapies. With NAD+ at the forefront, we may be on the cusp of a new era in cancer treatment, one that exploits the metabolic weaknesses of cancer cells to our advantage. The ultimate goal remains clear: to transform this metabolic vulnerability into a tangible benefit for cancer patients in the US and worldwide.









Comments